The National Agency for Food and Drug Administration and Control (NAFDAC) has warned Nigerians against the use of four substandard cough syrups.
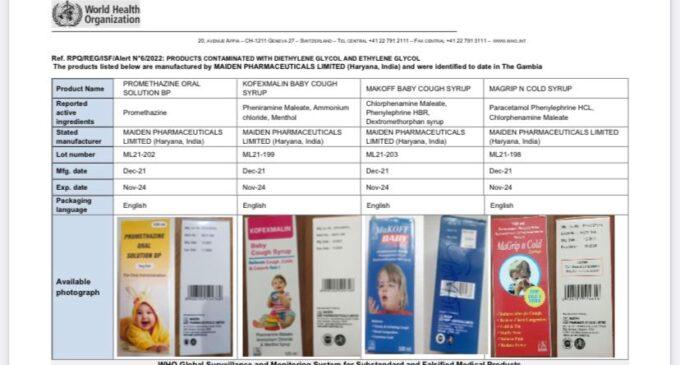
The products are listed as “Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup, and Magrip N Cold Syrup”.
The World Health Organisation (WHO) issued an alert on the syrups made by India-based Maiden Pharmaceuticals.
WHO had recently said the “contaminated medicines”, which were identified in Gambia, were “linked with acute kidney injuries and 66 deaths among children” in the country.
In a statement on Sunday, Mojisola Adeyeye, NAFDAC director-general, said all batches of the syrups should be considered unsafe.
According to NAN, she said the products are not registered by NAFDAC and should, therefore, not be in circulation.
Adeyeye implored importers, distributors, retailers and consumers to exercise caution and vigilance within the supply chain to avoid the importation, distribution, sale and use of substandard cough syrups.
She also advised members of the public in possession of the listed products to discontinue their sale or use and submit their stock to the nearest NAFDAC office.
“If you have these substandard products, please do not use them. If you or someone you know have used them or suffered any adverse reaction/event, you are advised to seek immediate medical advice from a qualified healthcare professional,” the statement reads.
The director general encouraged healthcare professionals and consumers to report any suspicion of adverse drug reactions and substandard medicines to NAFDAC via 0800-162-3322.
“The public is also advised to report any incident through this email: sf.alert@nafdac.gov.ng,” the statement reads.